EQUILIBRIUM & ACID Reactions
Inquiry question: What happens when chemical reactions do not go through to completion?
Students:
FACTORS THAT AFFECT EQUILIBRIUM
Inquiry question: What factors affect equilibrium and how?
Students:
CALCULATING THE EQUILIBRIUM CONSTANT ( KEQ )
Inquiry question: How can the position of equilibrium be described and what does the equilibrium constant represent?
Students:
SOLUTION EQUILIBRIA
Inquiry question: How does solubility relate to chemical equilibrium?
Students:
Enthalpy (H) is a measure of the heat contentof a system. Absolute enthalpy cannot be measured. However, the change in enthalpy (∆H): the change in the heat content of system during a process, measured at constant pressure.
Δ𝐻 = 𝐻𝑝𝑟𝑜𝑑𝑢𝑐𝑡𝑠 − 𝐻𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠
ENERGY PROFILE DIAGRAMS
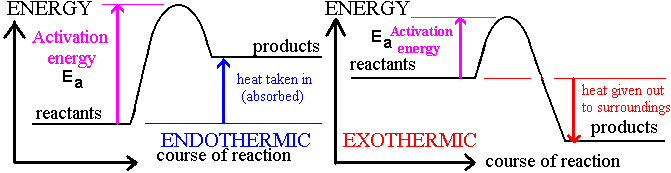
‘The total enthalpy change in a chemical reaction is constant, whether the reaction is performed in one step or
several steps.’
Hess’s law is a form of the law of conversation of energy (First law of Thermodynamics).

Bond energy (or bond enthalpy) is the amount of energy required to break one mole of a bond in a gaseous molecule.
∆𝐻𝑟𝑥𝑛 = ∑ ∆𝐻𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡 𝑏𝑜𝑛𝑑𝑠 𝑏𝑟𝑜𝑘𝑒𝑛 + ∑ ∆𝐻𝑝𝑟𝑜𝑑𝑢𝑐𝑡 𝑏𝑜𝑛𝑑𝑠 𝑓𝑜𝑟𝑚𝑒𝑑
∆𝐻𝑟𝑥𝑛 = ∑ ∆𝐻𝑏𝑜𝑛𝑑 𝑒𝑛𝑡ℎ𝑎𝑙𝑝𝑦 𝑜𝑓 𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠 − ∑ ∆𝐻𝑏𝑜𝑛𝑑 𝑒𝑛𝑡ℎ𝑎𝑙𝑝𝑦 𝑜𝑓 𝑝𝑟𝑜𝑑𝑢𝑐𝑡𝑠
Entropy (S) is a measure of how the available energy is distributed or dispersed amount particles in a system. It is also a measure of energy dispersal (function of temperature). Generally, low entropy à high entropy. (Chaos)
When energy can be distributed in more ways, there is a greater entropy
FACTORS THAT CHANGE ENTROPY
o The larger the volume, the more ways there are to distribute the energy
SECOND LAW OF THERMODYNAMICS
The second law of thermodynamics states that the entropy of the universe is always increasing.
Δ𝑆𝑢𝑛𝑖𝑣𝑒𝑟𝑠𝑒 = Δ𝑆𝑠𝑦𝑠𝑡𝑒𝑚 + Δ𝑆𝑠𝑢𝑟𝑟𝑜𝑢𝑛𝑑𝑖𝑛𝑔𝑠 > 0
CALCULATING ENTROPY IN CHEMICAL REACTIONS
Δ𝑆𝑟𝑥𝑛 = ∑ Δ𝑆𝑝𝑟𝑜𝑑𝑢𝑐𝑡𝑠 − ∑ Δ𝑆𝑟𝑒𝑎𝑐𝑡𝑎𝑛𝑡𝑠
In any process, the main form of interaction between the system and the surroundings is the exchange of heat.
The equation allows the comparison between the relative contributions of the two driving forces for a reaction, entropy and enthalpy.
- If 𝚫𝑮 < 𝟎 (Δ𝐻𝑠𝑦𝑠𝑡𝑒𝑚 < 𝑇Δ𝑆𝑠𝑦𝑠𝑡𝑒𝑚), a reaction is spontaneous
- If 𝚫𝑮 > 𝟎 (Δ𝐻𝑠𝑦𝑠𝑡𝑒𝑚 > 𝑇Δ𝑆𝑠𝑦𝑠𝑡𝑒𝑚), a reaction is non-spontaneous
-If 𝚫𝑮 = 𝟎, a reaction will occur both in the forward and reverse directions, equilibrium.

For reversible reactions, a reversible arrow is used to indicate that both reactions are capable of proceeding.
STATIC AND DYNAMIC EQUILIBRIUM
Reactions will proceed until either a static or dynamic equilibrium is reached. Equilibrium refers to the state of a
closed chemical system which:
Irreversible reactions (shown with a forward arrow →) that go to completion reach a static equilibrium.
Reversible reactions (shown with a reversible arrow ⇋) do not go to completion. In a closed system, reversible reactions will instead reach a state known as dynamic equilibrium.
There are no macroscopic changes when a closed system is at equilibrium.
TYPES OF SYSTEMS
RATES OF REACTION
In order for any reaction to proceed, reactants must collide. Particles need to collide with sufficient energy and in the correct orientation for it to be a successful reaction. A collision with sufficient energy and the correct orientation is called an effective collision.
The rate of reaction is how rapidly a reaction proceeds. The rate is defined as the change in the concentration of reactants or products over time. It is dependant on the frequency of effective collisions.
ADDITION/REMOVAL OF A REACTION COMPONENT
If the concentration of one reaction component increases, its rate of reaction will increase as there are more particles to collide with, thus increasing the frequency of effective collisions. The rate of that reaction will be relatively greater than that of the rate of the reverse reaction. This means that more products or reactants will being produced until equilibrium is reached.
Similarly, the reduction in one of the reaction components will reduce its rate of reaction. This occurs as there are fewer particles to collide with which reduces the frequency of effective collisions. The rate of that reaction will be relatively less than the rate of the reverse reaction. This means that more products or reactants will being produced until equilibrium is reached.
CHANGE IN VOLUME OR PRESSURE
A decrease in the volume of a chemical system involving gasses will result in gasses colliding more often. The gas particles will also be colliding with more energy as pressure is inversely proportional to volume. As particles are colliding more frequently and with more energy to overcome the activation energy barrier, the frequency of effective collision increases. Both the rate of the forward and reverse reaction will increase, however, the rate of reaction that uses the greatest number of moles will be relatively greater than the reverse reaction as there are more particles that can collide effectively with each other.
When the volume is increased and pressure is decreased, the partial pressures of all gasses will decrease. Both the rate of the forward and reverse reaction will decrease. The rate of reaction that produces more moles will be relatively greater than the reverse as the reaction is more likely to occur because it requires fewer particles to effectively collide.
Changing the overall pressure of a chemical system does not always cause a disturbance in equilibrium. For example, the addition of inert gasses.
CHANGE IN TEMPERATURE
By increasing the temperature of a chemical system at equilibrium, particles will possess more kinetic energy, which means that more particles (both reactants and products) have enough energy to collide and overcome the activation energy of the forward and reverse reactions.
For an exothermic reaction, the activation energy of the reverse reaction is higher than that of the forward reaction. An increase in temperature means that proportionally more products will be able to collide with enough energy in reverse reaction than the reactants. This causes the rate of the reverse reaction to occur at a faster rate than the forward reaction. Therefore, the concentrations of the reactants will increase whereas the concentration of the products will decrease until a new state of equilibrium is reached.
When the temperature is decreased, all the particles in the system lose energy which decreases both the rate of the forward and the reverse reaction. However, the rate of the reverse endothermic reaction will be relatively higher than the rate of the forward reaction.
OUTCOMES COVERED
- combustionreactions
- photosynthesis
Photosynthesis appears to be the reverse reaction of the combustion of glucose and may seem to be a reversible reaction. However, in nature, the process involves many individual irreversible steps which combine to give the overall reaction, hence photosynthesis is irreversible.
6CO2(g) + 6H2O(l) → C6H12O6(s) + 6O2(g)
-∆𝐻 > 0 (endothermic)
-∆𝑆 > 0 (less moles, moving to more order)
-Non-spontaneous at all temperatures
-Chlorophyll is the catalyst for photosynthesis.
-UV rays drives the photosynthesis reaction.
The sign of ∆𝐺 indicates whether reactants or products will dominate the mixture with lowest free energy.
OUTCOMES COVERED
FACTORS AFFECTING THE RATE OF REACTION
NATURE OF REACTANTS
Every reaction has its own rate and its own activation energy, depending on the reactivity of the reactants. Aqueous solutions already have dissociated ions. They do not need to collide in any correct orientation and usually have very low Ea.
CONCENTRATION
The rate of reaction increases when the concentration of reactants is increased.
SURFACE AREA (PARTICLE SIZE)
The rate of reaction increases when the surface area of reactants is increased.
TEMPERATURE
The rate of reaction increases when the temperature of the reactants is increased.
PRESENCE OF A CATALYST
A catalyst is a substance that increases the rate of reaction without being consumed. Catalysts work by allowing the reaction to take an alternative reaction pathway with a lower activation energy
In 1888, a French chemist called Henri Le Chatelier (1850 1936) put forth the statement known as Le Chatelier’s.
Principle:
“If a system at dynamic equilibrium is disturbed by changing the conditions, the system undergoes a reaction which minimises the effect of the disturbance to attain a new equilibrium”
A chemical system at equilibrium can be disturbed in the following ways:
Le Chatelier’s principle is a convenient method for predicting equilibrium shifts, but does not explain why it shifts. Collision theory explains the shift in equilibrium.
CHANGE IN CONCENTRATION
N2(g) + 3H2(g) ⇋ 2NH3(g) ∆𝐻 = −92𝑘𝐽 𝑚𝑜𝑙−1
ADDING A REACTION COMPONENT
Addition of H2(g) increases [H2(g)]. This will result in the rate of the forward reaction to increase, meaning the forward reaction has been favoured.
Since the rate of the forward reaction is different to the rate of the reverse reaction, the equilibrium has been disturbed.
Generally, the reaction that counteracts the disturbance will be favoured; in other words, its rate will increase relative to the other reaction.
REMOVING A REACTION COMPONENT
Removing N2(g) decreases [N2(g)]. This will result in the rate of the reverse reaction to increase, meaning the reverse reaction has been favoured.
The reaction that counteracts the disturbance will be favoured; in other words, its rate will increase relative to the other reaction.
CHANGE IN VOLUME (OR PRESSURE)
The pressure exerted by a gas arises from the force of the gas particles colliding with the walls of the container. Therefore, the pressure is proportional to the number of gas particles present.
Boyle’s Law states that pressure and volume are inversely proportional to each other.
CHANGE IN PRESSURE
Doubling the pressure of the container decreases volume. To counteract this effect, the reaction will shift in the direction that produces the least amount of moles. Therefore, the forward reaction will be favoured.
CHANGE IN TEMPERATURE
The effect of a change in temperature on a reaction at equilibrium depends on whether the forward reaction is exothermic or endothermic.
If temperature of the system is increased, the reverse reaction will be favoured to counteract this effect. The reverse reaction is endothermic and will be favoured.
ADDITION OF A CATALYST
A catalyst increases the rate of a chemical reaction without being consumed, by providing an alternate pathway of lower activation energy. The addition of a catalyst reduces the activation energy of both the forward and reverse reaction by the same amount.
ADDITION OF INERT GAS
The addition of an inert gas will increase pressure, but equilibrium will not be disturbed. This is because the concentrations of reactants and products remain the same, if the volume of the container does not change.
When colourless dinitrogen tetroxide gas (N2O4) is enclosed in a vessel, a brown colour will appear indicating the formation of nitrogen dioxide (NO2). The intensity of the brown colour indicates the amount of nitrogen dioxide present in the vessel. The dimerization of nitrogen dioxide is an exothermic process
NOTES
Clearly explain the effect of changes in conditions on the yield of equilibrium reactions.
OUTCOMES
Investigatethe effects of temperature, concentration, volume and/or pressure on a system at
equilibrium and explain how Le Chatelier’s principle can be used to predict such effects, for example:
The changes occurring in a system can be identified by examining the shape of the line in a concentration profile diagram during a particular time period.
OUTCOMES
A quantitative way of describing the position of equilibrium is the equilibrium constant:
𝑎𝐴 + 𝑏𝐵 ⇋ 𝑐𝐶 + 𝑑𝐷
In an ideal system, the value of K is constant at constant temperature.
PURE, LIQUIDS AND SOLIDS
𝐶𝑎𝐶𝑂3(𝑠) ⇋ 𝐶𝑎𝑂(𝑠) + 𝐶𝑂2(𝑔)
The concentrations of pure solids and pure liquids cannot change at a constant temperature.
OUTCOMES
The larger the value of K, the further the equilibrium lies towards the RHS. A reaction with a very large K, proceeds almost to completion.
The smaller the value of K, the further the equilibrium lies towards the LHS. A reaction with a very small K, proceeds barely at all.
K AND THE DIRECTION OF REACTION
If concentrations are substituted into the expression at any point, the value is called Q, the reaction quotient.
The relative values of Q and K determines which way the reaction will proceed to reach equilibrium.

CALCULATIONS WITH QUADRATIC EQUATIONS
In some calculations, it will be required to solve a quadratic equation to determine 𝑥.
In these calculations, simplification can be used to make the calculation less complicated to solve:
OUTCOMES
When temperature is increased, equilibrium shifts so that the endothermic reaction is favoured, whether it is the forward or the reverse reaction. Therefore, unlike changes in concentration and pressure, a change in temperature is the only factor that will change the value of K.
For exothermic reactions, K increases with lower temperatures and decreases with higher temperatures.
For endothermic reactions, K decreases with lower temperatures and increases with higher temperatures.
OUTCOMES
IRON (III) THIOCYANATE EQUILIBRIUM REACTION
When mixed together, aqueous solutions of iron(III) nitrate (Fe(NO3)3) and potassium thiocyanate (KSCN) combine, in a reversible exothermic process, to form the aqueous iron(III) thiocyanate complex ([Fe(SCN)]2+ ).
Iron(III) ions are very pale yellow colour, while iron(III) thiocyanate complex is an intense deep red colour. When diluted and at equilibrium, the colour of the mixture containing all three species is amber.
EQUILIBRIUM SHIFTS
A shift to the RHS, increase concentration of [Fe(SCN)]2+ → More intense
A shift to the LHS, decreases concentration of [Fe(SCN)]2+ → Less intense
NOTES
Since thiocyanate ions bind to iron via the nitrogen atom, the formula of the iron(III) thiocyanate complex is sometimes written as [Fe(NCS)]2+.
WHAT IS SPECTROPHOTOMETRY
Spectrophotometry is an analytical technique that is used to determine the concentration of a substance in a solution.
A UV/Vis spectrophotometer like the one shown measure the intensity of light passing through a sample solution in a cuvette.
A spectrophotometer expresses the intensity of light in absorbance (A).
Absorbance is proportional to both concentration and the length of the sample, according to the Beer-Lambert Law.
A process where equilibrium is often established is in the dissolution of ionic compounds in water.
- Brittle
- HighMP, BP
- Solidsat room temperature
- Moltenand aqueous state conducts electricity
SOLUTIONS OF IONIC COMPOUINDS
Soluble ionic compounds will dissolve in water to form aqueous solutions. When a soluble ionic compound is added to water, the ions at the surface of the crystal become surrounded by water molecules.
o They form ion-dipole forces between the molecules.
When the ion-dipole forces between the ions and the permanent dipoles of the water molecules (adhesive forces) become stronger than the ionic bonds between the ions and the hydrogen bonding within the water (cohesive forces), the ions are dislodged from their position in the crystal.
The solvated or hydrated ions are surrounded by a shell of water molecules known as a solvation layer.
Many ionic compounds are soluble in water and will dissociate to form aqueous solutions.
Entropy can also contribute to solubility. Most dissolutions are entropically favourable (∆𝑆 > 0), but some dissolutions are unfavourable.
∆𝑆𝑠𝑜𝑙𝑢𝑡𝑒 + ∆𝑆𝑠𝑜𝑙𝑣𝑒𝑛𝑡 = 𝑜𝑣𝑒𝑟𝑎𝑙𝑙 ∆𝑆
INCREASE IN ENTROPY
Solute in a solid state has a fixed ordered arrangement. Dissolved solute has free mobile ions. Therefore, there is an increase in entropy as the number of possible arrangement increases.
DECREASE IN ENTROPY
Pure water molecules are in random arrangements and are also mobile. When water becomes a solvent, molecules solvate the solute and has less possible arrangements decreasing entropy.
NOTES
“Clear” means that light can pass through the substance without being scattered.
“Colourless” means the substance is not coloured. Solutions are clear, but not necessarily colourless.”
OUTCOMES
conduct an investigation to determine solubility rules, and predict and analyse the composition of substances when two ionic solutions are mixed, for example:
PRECIPITATION
The production of an insoluble compound, usually by reacting two soluble compounds.
NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq)
When two clear solutions are mixed together, an insoluble compound is formed. This is called a precipitate reaction. Precipitation reactions are used to remove minerals from drinking water, to remove heavy metals from wastewater and in purification plants of reservoirs.
SOLUBILITY RULES
OUTCOMES
The solubility of a compound is the maximum amount of solute that can dissolve in a specific volume of solvent at a certain temperature.
EQUILIBRIA IN SATURATED SOLUTIONS
When a solvent has dissolved all the solute it can at a given temperature, the resulting solution is saturated.
In a saturated solution, the system is in dynamic equilibrium:
The equilibrium constant for these solution equilibria is called the solubility product constant (Ksp).
MOLARITY
Molarity is the main unit of concentration used in chemistry:
-Thevolume of solution is expressed in litres (L).
PERCENTAGE BY MASS OR WEIGHT (% M/M OR % W/W)
PERCENTAGE BY VOLUME (% V/V)
MASS PER VOLUME (M/V)
-A blood alcohol level of 0.01 refers to 0.010/g / 100mL of blood
PARTS PER MILLION & PARTS PER BILLION
Partsper million and parts per billion (ppm and ppb) are useful when describing very dilute
SUMMARY
OUTCOMES
Many native foods eaten by Aboriginal and Torres Strait Islander people are poisonous and need to be detoxified before consumption
There are several physical and chemical processes user for detoxification:
COOKING OR ROASTING (CHEMICAL REACTION)
LEACHING (PHYSICAL CHANGE)
FERMENTATION OR PROLONGED STORAGE (CHEMICAL REACTION)
They are digested by fungi, or broken down by the plant's natural enzymes. (Biological catalyst)
Cycads are palm-like plants that produce seeds in cones. Cycad seeds are a rich source of carbohydrates and have been eaten in regions of Northern Australia for thousands of years. However, the seeds contains highly toxic chemicals.
The two main types are cycasin and b-methylamino-l-alanine (BMAA).
To prepare them, Aboriginal and Torres Strait Islander people who ate these seeds would commonly prepare them by:
THE POISON, CYCASIN
SOLUBILITY
DECOMPOSITION
MORETON BAY CHESTNUT (BLACK BEAN)
The Moreton Bay Chestnut is found on the east coast of Australia. It produces pods containing large seeds that are toxic. Eating unprocessed seeds causes vomiting and diarrhoea. When cooked, processed seeds taste like sweet chestnuts.
OUTCOMES
CALCULATING 𝑲𝒔𝒑 FROM SOLUBILITY
An equilibrium is only present in a saturated solution where the maximum amount of ionic compound has dissolved. Therefore, the solubility of a substance, whether given in moles per litre or mass per volume, can be used to calculate the solubility constant 𝐾𝑠𝑝.
PREDICTING THE FORMATION OF A PRECIPITATE
Solubility constants can be used to predict if a precipitate will form when two solutions are mixed.
If the concentration of Ba2+ and OH- are higher than the amounts in a saturated solution, 𝑄 > 𝐾𝑠𝑝 and precipitation
will occur.
If the concentration of Ba2+ and OH- are lower than the amounts in a saturated solution, 𝑄 < 𝐾𝑠𝑝 and precipitation will not occur.
In an aqueous solution of an ionic compound, the ions are dissociated.
This means that in a saturated solution, if another substance is added that has an ion in common with the first substance, it will affect the position of equilibrium, leading to lower solubility. This is known as the common ion effect.
Since dissolution is a reversible process, the position of equilibrium (extent of dissolution) will depend on temperature.
The relationship between solubility and temperature can be seen on a solubility curve, which shows the maximum amount of solute that can dissolve at a range of temperatures.
OUTCOMES
– dissociation of acids and bases (ACSCH098, ACSCH099)

Acids differ in their strength: the extent of ionisation or dissociation in water.
- A stronger acid will ionise further
Strong acids completely dissociate in water
- Straight arrows are used to indicate that these dissociations are irreversible and proceed to completion.
Weak acids partially dissociate in water.
- Reversible arrows are used indicate that these dissociations proceed to equilibrium.
The acid dissociation constant is the equilibrium constant for the dissociation (ionisation) of an acid into hydrogen
ions (H+) and an anion (Ka).
Acids that can produce more than one H+ ion are known as polyprotic acids.
Acid strength depends on the identity of the acid and the extent of its ionisation in water.
STRONG ACIDS
- Perchloric: HClO4
- Hydriodic: HI
- Hydrobromic: HBr
- Hydrochloric: HCl
- Nitric: HNO3
- Sulfuric: H2SO4
The acidity of a solution is determined by both the strength and the concentration of the acids present.
The concentration of the hydrogen ions in a solution are generally small. The pH scale, a logarithmic scale, is a
convenient way of expressing [H+] as a number generally between 0 (extremely acidic) and 14 (extremely basic).
A pH of 7 represents a neutral solution. The further away from 7, the more acidic or alkaline the solution.
pH can be calculated from the [H+] using the equation:
pH = -[H+]
- p stands for −log10, and the concentration of the hydrogen ions are in mol L−1
- The notation pH derives from the French pouvoir hydrogene, meaning the “power of hydrogen”
To calculate the percentage of any component in a sample, the formula is:
FOR A WEAK ACID HA: